Sustained Weight Loss
In clinical trials, Saxenda® helped adult patients maintain weight loss1
Actor portrayal.
Clinically Meaningful Weight Loss
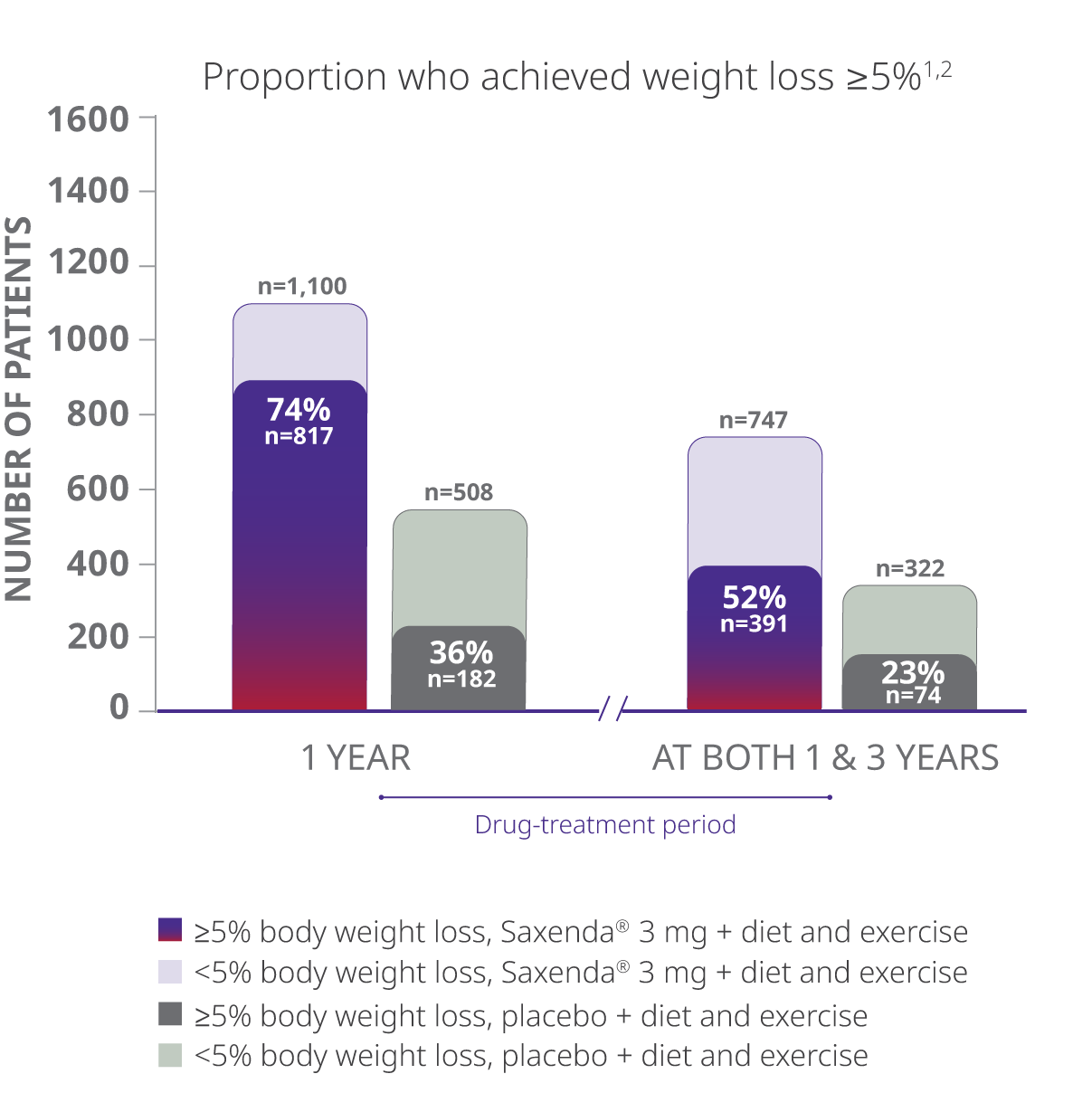
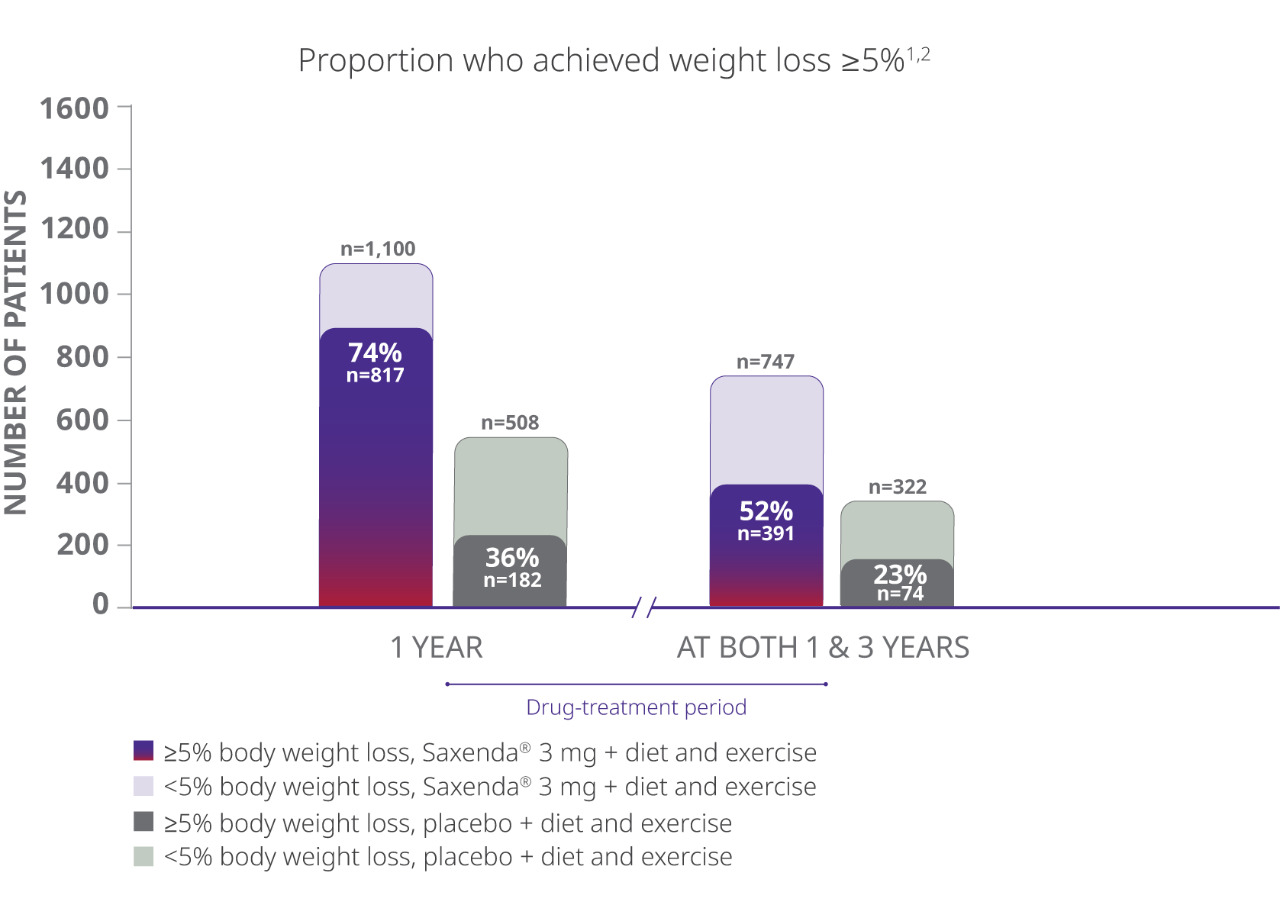
In a 3-year study in adults, more than 50% of patients taking Saxenda® who completed the trial achieved clinically meaningful weight loss at year 1 and maintained it at 3 years1,2
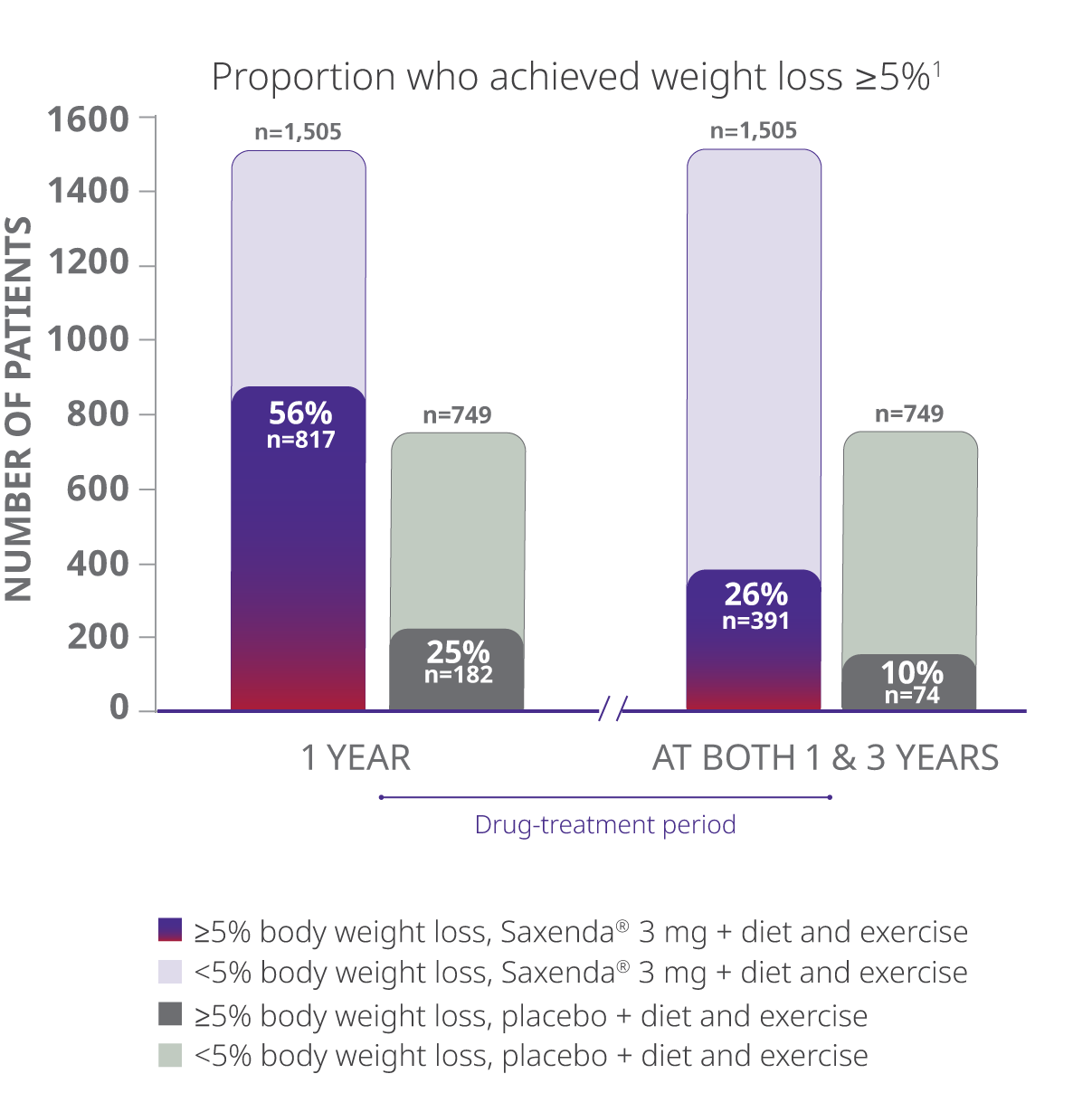
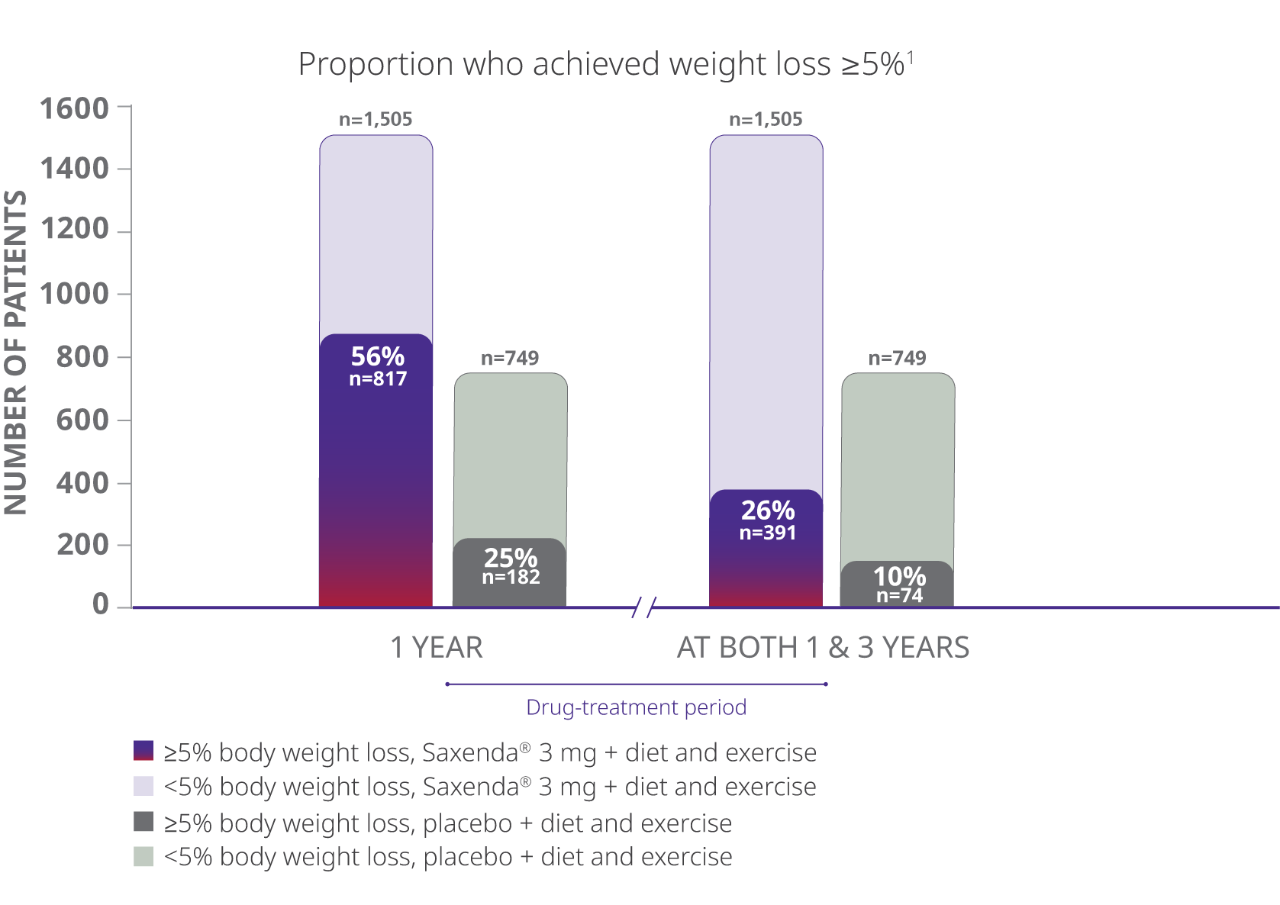
Of the total randomized population, 26% of patients taking Saxenda® achieved clinically meaningful weight loss at year 1 and maintained it at year 3
In a 3-year study of 2,254 adult patients with pre-diabetes and with a BMI of either ≥30 kg/m2, or ≥27 with at least 1 additional comorbidity, all patients received a reduced-calorie diet (~500-kcal/day deficit) and physical activity counseling. Trial did not stipulate assessing weight at 3 years for those patients who did not complete the trial.1
Total Randomized Trial Population
Completer Population
Upon discontinuation of treatment, patients are likely to regain weight3-5
- 53% of Saxenda® patients and 45% of patients on placebo completed the 3-year trial3
- The safety profile over 3 years was consistent with the profile at 1 year3
Study 1 (3 year)1,3
- Results from a 160-week, randomized, double-blind, placebo-controlled study to evaluate the long-term safety and efficacy of Saxenda®
- Adult patients with pre-diabetes and with a BMI of either ≥30, or ≥27 with at least 1 additional comorbidity, were randomized to receive once-daily Saxenda® (n=1,505) or placebo (n=749) in conjunction with a lifestyle modification program that included increased physical activity and a 500-kcal/day-deficit diet
- Patients underwent a 4-week dose-escalation period followed by 156 weeks on the full dose, with a 12-week off-drug observational follow-up period
- The study evaluated percentage of patients achieving weight loss of at least 5% of body weight at both 1 year and 3 years
- Mean baseline body weight was 236.7 lb and mean BMI was 38.8 kg/m2
In patients with obesity, losing weight causes changes in appetite hormones that can actually increase hunger and the desire to eat.6
That's why it is important to design a weight-management plan that focuses on not only achieving but maintaining clinically meaningful weight loss.
RECOMMENDED CONTENT
Additional Weight Loss
Important Safety Information for Saxenda® (liraglutide) injection 3 mg
WARNING: RISK OF THYROID C-CELL TUMORS
Liraglutide causes dose-dependent and treatment-duration-dependent thyroid C-cell tumors at clinically relevant exposures in both genders of rats and mice. It is unknown whether Saxenda® causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans, as the human relevance of liraglutide-induced rodent thyroid C-cell tumors has not been determined.
Saxenda® is contraindicated in patients with a personal or family history of MTC and in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Counsel patients regarding the potential risk of MTC with use of Saxenda® and inform them of symptoms of thyroid tumors (eg, a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with Saxenda®.
Indications and Usage
Saxenda® (liraglutide) injection 3 mg is indicated as an adjunct to a reduced-calorie diet and increased physical activity for chronic weight management in:
- Adult patients with an initial body mass index (BMI) of 30 kg/m2 or greater (obese) or 27 kg/m2 or greater (overweight) in the presence of at least one weight-related comorbid condition (eg, hypertension, type 2 diabetes mellitus, or dyslipidemia)
- Pediatric patients aged 12 years and older with body weight above 60 kg (132 lbs) and initial BMI corresponding to 30 kg/m2 or greater for adults (obese) by international cut-offs
Limitations of Use
- Saxenda® contains liraglutide and should not be coadministered with other liraglutide-containing products or with any other GLP-1 receptor agonist
- The safety and effectiveness of Saxenda® in pediatric patients with type 2 diabetes have not been established
- The safety and effectiveness of Saxenda® in combination with other products intended for weight loss, including prescription drugs, over-the-counter drugs, and herbal preparations, have not been established
Important Safety Information cont.
Contraindications
Saxenda® is contraindicated in:
- Patients with a personal or family history of MTC or patients with MEN 2
- Patients with a serious hypersensitivity reaction to liraglutide or to any of the excipients in Saxenda®. Serious hypersensitivity reactions including anaphylactic reactions and angioedema have been reported with Saxenda®
- Pregnancy
Warnings and Precautions
- Risk of Thyroid C-cell Tumors: If serum calcitonin is measured and found to be elevated, the patient should be further evaluated. Patients with thyroid nodules noted on physical examination or neck imaging should also be further evaluated
- Acute Pancreatitis: Acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis, has been observed in patients treated with liraglutide postmarketing. Observe patients carefully for signs and symptoms of pancreatitis (persistent severe abdominal pain, sometimes radiating to the back with or without vomiting). If pancreatitis is suspected, discontinue Saxenda® promptly and if pancreatitis is confirmed, do not restart
- Acute Gallbladder Disease: Substantial or rapid weight loss can increase the risk of cholelithiasis; however, the incidence of acute gallbladder disease was greater in patients treated with Saxenda® than with placebo even after accounting for the degree of weight loss. If cholelithiasis is suspected, gallbladder studies and appropriate clinical follow-up are indicated
- Hypoglycemia: Adult patients with type 2 diabetes on an insulin secretagogue (eg, a sulfonylurea) or insulin may have an increased risk of hypoglycemia, including severe hypoglycemia with use of Saxenda®. The risk may be lowered by a reduction in the dose of insulin secretagogues or insulin. In pediatric patients without type 2 diabetes, hypoglycemia occurred. Inform all patients of the risk of hypoglycemia and educate them on the signs and symptoms
- Heart Rate Increase: Mean increases in resting heart rate of 2 to 3 beats per minute (bpm) were observed in patients treated with Saxenda®. Monitor heart rate at regular intervals and inform patients to report palpitations or feelings of a racing heartbeat while at rest during treatment with Saxenda®. Discontinue Saxenda® in patients who experience a sustained increase in resting heart rate
- Renal Impairment: Acute renal failure and worsening of chronic renal failure, which may sometimes require hemodialysis, have been reported, usually in association with nausea, vomiting, diarrhea, or dehydration. Use caution when initiating or escalating doses of Saxenda® in patients with renal impairment
- Hypersensitivity Reactions: Serious hypersensitivity reactions (eg, anaphylaxis and angioedema) have been reported in patients treated with Saxenda®. If a hypersensitivity reaction occurs, patients should stop taking Saxenda® and promptly seek medical advice
- Suicidal Behavior and Ideation: In adult clinical trials, 9 (0.3%) of 3,384 patients treated with Saxenda® and 2 (0.1%) of the 1,941 treated with placebo reported suicidal ideation; one of the Saxenda® treated patients attempted suicide. In a pediatric trial, 1(0.8%) of the 125 Saxenda® treated patients died by suicide. There was insufficient information to establish a causal relationship to Saxenda®. Monitor patients for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior. Discontinue treatment if patients experience suicidal thoughts or behaviors. Avoid Saxenda® in patients with a history of suicidal attempts or active suicidal ideation
Adverse Reactions
- The most common adverse reactions, reported in ≥5% are nausea, diarrhea, constipation, vomiting, injection site reactions, headache, hypoglycemia, dyspepsia, fatigue, dizziness, abdominal pain, increased lipase, upper abdominal pain, pyrexia, and gastroenteritis
Drug Interactions
- Saxenda® causes a delay of gastric emptying and has the potential to impact the absorption of concomitantly administered oral medications. Monitor for potential consequences of delayed absorption of oral medications concomitantly administered with Saxenda®
Use in Specific Populations
- There are no data on the presence of liraglutide in human breast milk; liraglutide was present in the milk of lactating rats
- Saxenda® has not been studied in patients less than 12 years of age
- Saxenda® slows gastric emptying. Saxenda® has not been studied in patients with preexisting gastroparesis
Please click here for Saxenda® Prescribing Information, including Boxed Warning.
References
1. Saxenda® [package insert]. Plainsboro, NJ: Novo Nordisk Inc.; 2022.
2. Data on file. Novo Nordisk Inc.; Plainsboro, NJ.
3. le Roux CW, Astrup A, Fujioka K, et al. 3 years of liraglutide versus placebo for type 2 diabetes risk reduction and weight management in individuals with prediabetes: a randomised, double-blind trial. Lancet. 2017;389(10077):1399-1409.
4. Wadden TA, Hollander P, Klein S, et al.; For NN8022-1923 Investigators. Weight maintenance and additional weight loss with liraglutide after low-calorie-diet-induced weight loss: the SCALE Maintenance randomized study. Int J Obes (Lond). 2013;37(11):1443-1451.
5. Davies MJ, Bergenstal R, Bode B, et al.; For NN8022-1922 Study Group. Efficacy of liraglutide for weight loss among patients with type 2 diabetes: the SCALE Diabetes randomized clinical trial. JAMA. 2015;314(7):687-699.
6. Sumithran P, Prendergast LA, Delbridge E, et al. Long-term persistence of hormonal adaptations to weight loss. N Engl J Med. 2011;365(17):1597-1604.